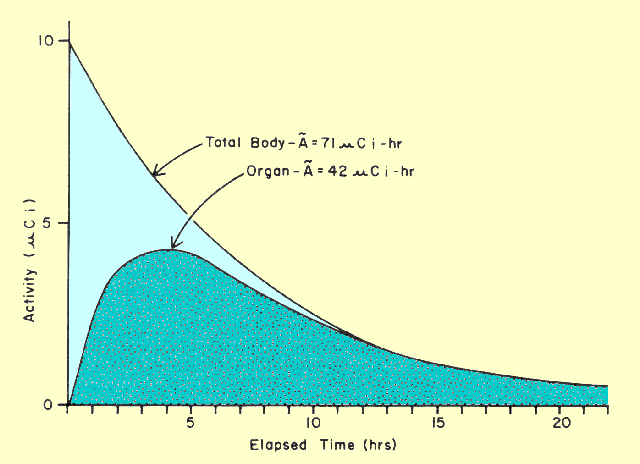
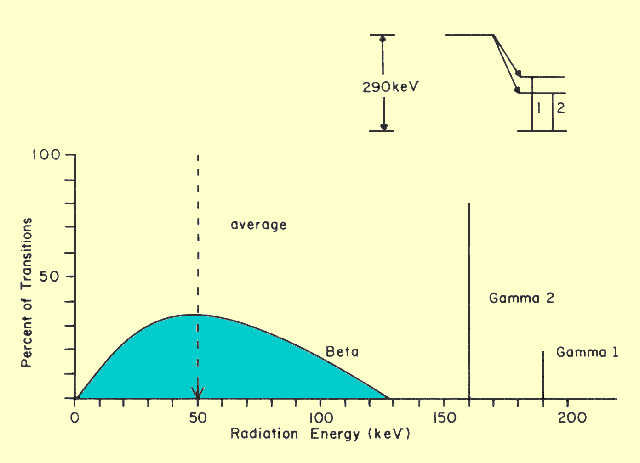
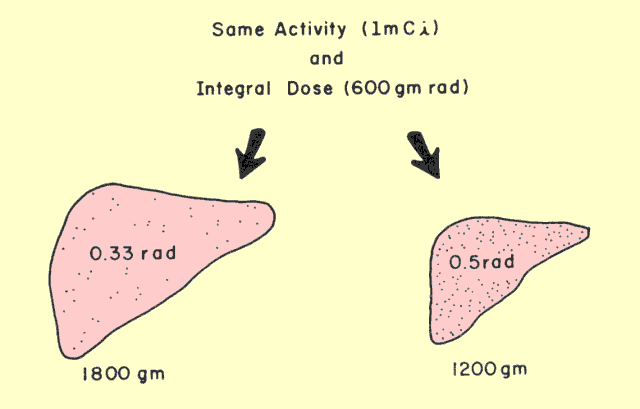
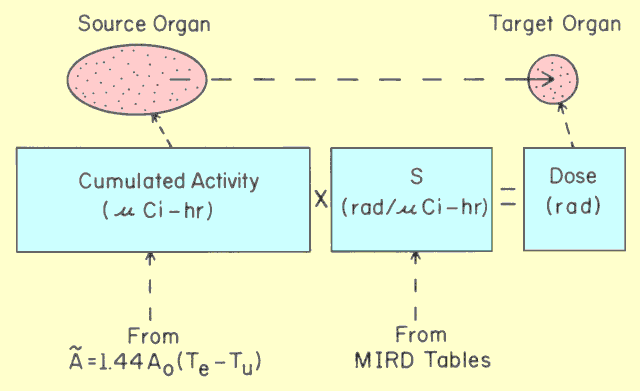
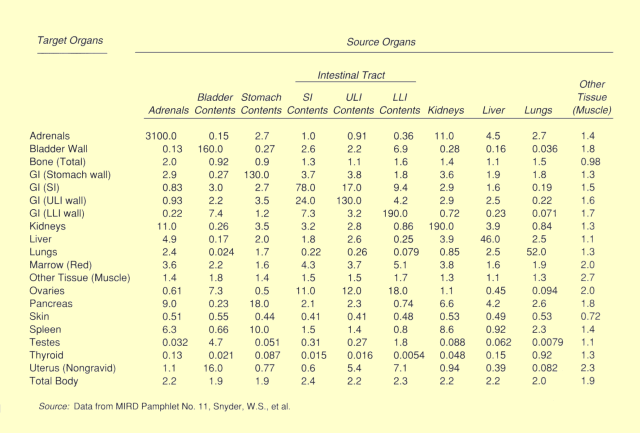
|
|||||
|
|
|||||
INTRODUCTION AND OVERVIEW |
|||||
|
All medical imaging methods deposit some form of energy in the
patient's body. Although the quantity of energy is relatively low, it
is a factor that should be given attention when conducting diagnostic
examinations. In general, there is more concern for the energy
deposited by the ionizing radiations, x-ray and gamma, than for
ultrasound energy or radio frequency (RF) energy deposited in magnetic
resonance imaging (MRI) examinations. Therefore, this chapter gives
major emphasis to the issues relating to the exposure of patients to
ionizing radiation.
Patients undergoing either x-ray or radionuclide examinations are
subject to a wide range of exposure levels. One of our objectives is to
explore the factors that affect patient exposure. This is followed by
an explanation of methods that can be used to determine patient
exposure values in the clinical setting.
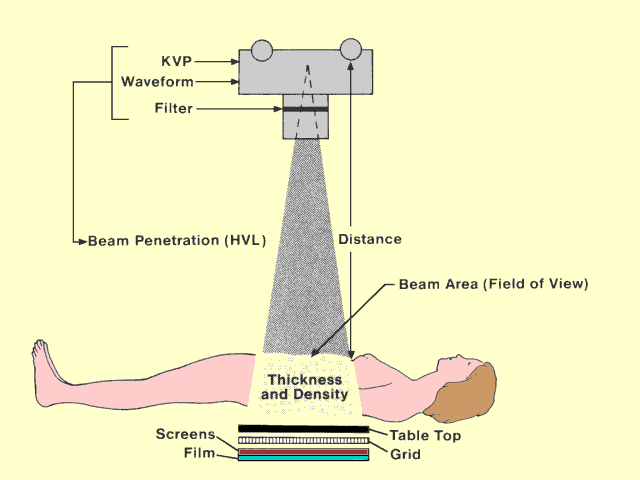
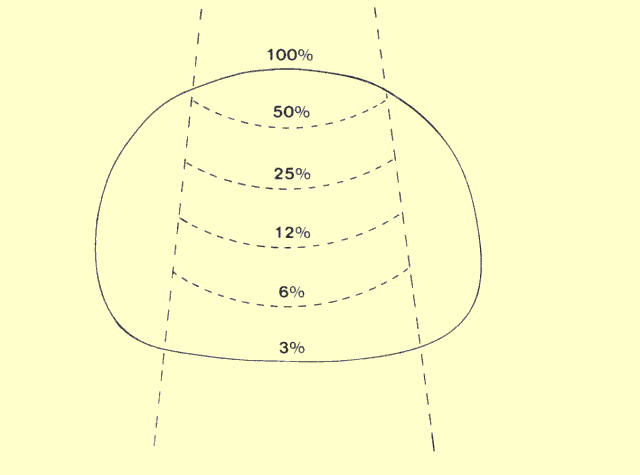
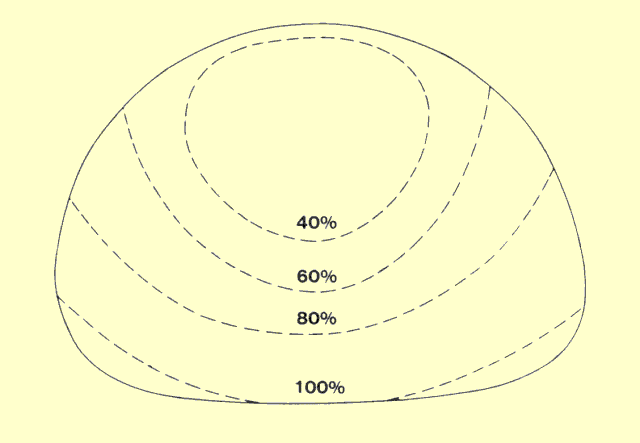
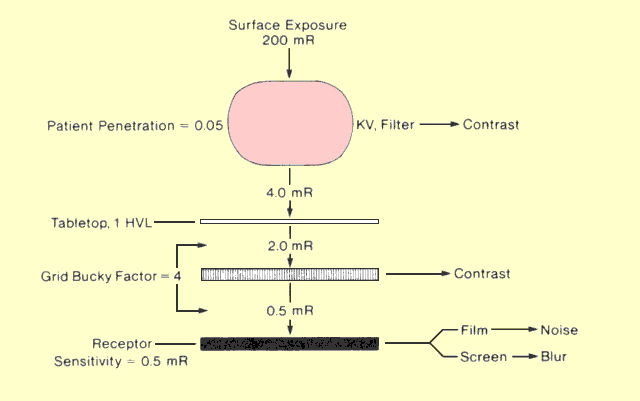
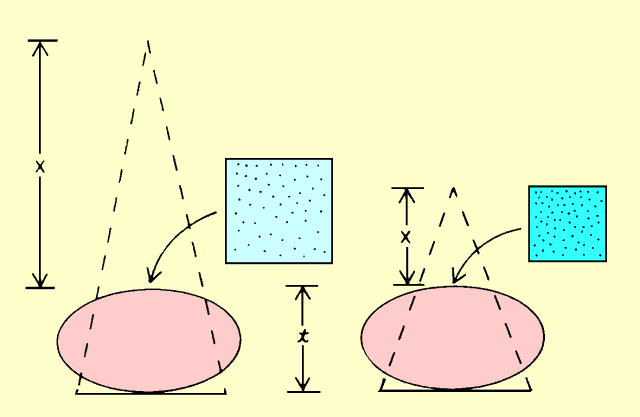
The following figure identifies the major factors that affect patient
exposure during a radiographic procedure. Some factors, such as
thickness and density, are determined by the patient. Most of the
others are determined by the medical staff. Many of the factors that
affect patient exposure also affect image quality. In most instances
when exposure can be decreased by changing a specific factor, image
quality is also decreased. Therefore, the objective in setting up most
x-ray procedures is to select factors that provide an appropriate
compromise between patient exposure and image quality.
Factors That Affect Patient Exposure in a Radiographic Procedure |
|||||